P629
Liposomes are synthetic vesicles bound by a phospholipid bilayer and capable of entrapping a variety of molecules. Liposomes consist of an aqueous core surrounded by a lipid bilayer, much like a membrane, separating the inner aqueous core from the bulk outside. They usually range in size from 50 nm to 200 nm, depending on the application.
A research group formulated a bone marrow-targeted stealth liposome that efficiently entrapped meglumine antimoniate, a drug used for treating leishmaniasis. These liposomes had a high entrapment capacity for water-soluble molecules and were composed of DPPC, cholesterol, PEG-DSPE and succinic acid-lipid conjugate (SA-lipid). The uptake of liposomes by bone marrow phagocytes in rhesus monkeys, rabbits, and hamsters was induced by the incorporation of SA-lipid. Grafting of PEG-DSPE enhanced the uptake of the liposomes by bone marrow phagocytes passively by preventing their uptake by hepatic and splenic phagocytes.
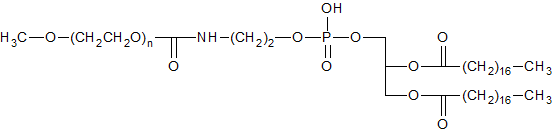
Figure 1. Structure of PEG-DSPE.
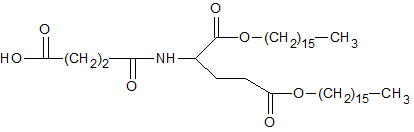
Figure 2. Structure of SA-lipid.
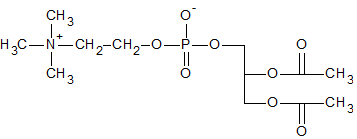
Figure 3. Structure of DPPC.
Liposomes can be assembled with a range of characteristics including different size, surface charge (cationic, anionic or neutral) and drug-to-lipid ratios. These properties can be tailored for a specific drug, specific application and the target site of delivery. There is a range of clinical products which exploits liposomes to carry drugs to the appropriate site of action, thereby improving specificity and reducing toxicity. Liposomes can also be actively targeted to specific cells or subcellular regions using targeting ligands attached to their surface.
.
Find an error? Take a screenshot, email it to us at error@mytestingsolution.com, and we’ll send you $3!
